 
This difference in formation leads to a difference in strength. Sigma bonds are formed by direct head to head overlap between orbitals while pi bonds are formed by side to side overlap, usually between p orbitals. stable than the atomic s and p orbitals, even tho formally, the why p-p sigma bond. The main differences between sigma and pi bonds has to do with their formation and strength. In cyclopropane it has been found that significant electron density lies off the internuclear axis, rather than along the axis.įurther, the $\ce$. Whereas pi bonds are formed in addition to sigma bonds by sideways. In general, single bonds between atoms are always sigma bonds. For a carbon with 1 double bond and 2 single bonds, the orbitals will become 33 's' and 66.7 'p' making it 'sp2. In order to better understand the bent bond model let's first consider its application to cyclopropane and then move to ethylene. (1) bonds result from end-to-end overlap of atomic orbitals (2) bonds result in the. Hückel's description is the one commonly seen in introductory texts, but both methods produce equivalent descriptions of the electron distribution in a molecule. Bonding: A chemical bond is formed between the ions or molecules due to the attraction. Bent bonds, tau bonds or banana bonds whatever you might like to call them were proposed by Linus Pauling Erich Hückel proposed the alternative $\sigma - \pi$ bonding formalism. Bonds formed from overlap of atomic s orbitals are always sigma bonds. There can be, even in simple carbon compounds. Remember that covalent bonds form from the overlap of atomic orbitals which are just the space where electrons are likely to be found.Why can there not be more than one sigma bond in a set of bonds? Saturated Unsaturated and Supersaturated.POSSIBLE ORBITAL COMBINATIONS TO GENERATE SIGMA MOLECULAR ORBITALS For simplicity, if we examine only the s, p, and d orbitals, let's suppose that all orbitals we are examining are similar enough in energy to interact. Reaction Quotient and Le Chatelier's Principle bonds must be made by orbitals that overlap head-on.Prediction of Element Properties Based on Periodic Trends.A sigma bond can be formed by overlap of an s atomic orbital with a p atomic orbital. Molecular Structures of Acids and Bases This type of bond is referred to as a (sigma) bond. 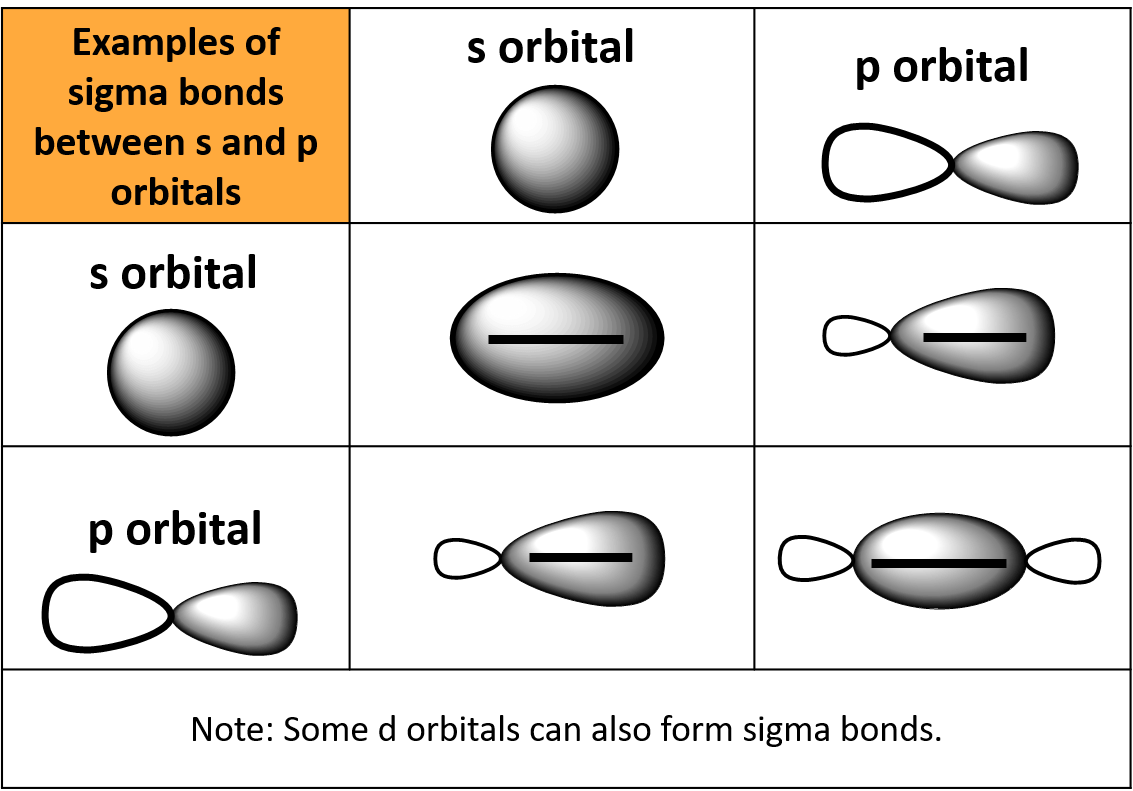
A Sigma bond is the single bond present between two atoms. Ion and Atom Photoelectron Spectroscopy Step3: For bonds formed from atomic s orbitals are always bonds.Elemental Composition of Pure Substances.Application of Le Chatelier's Principle.Structure, Composition & Properties of Metals and Alloys.Intramolecular Force and Potential Energy.Variable Oxidation State of Transition Elements.The type of chemical bond maximizes the stability of the atoms that form it. One of the bonds in a double or triple bond is always a sigma bond. Atoms form chemical bonds to make their outer electron shells more stable. Transition Metal Ions in Aqueous Solution Molecular orbitals are formed when the atomic orbitals of two (or more) different.Single and Double Replacement Reactions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
